Abstract
Background: The Chimeric antigen receptor (CAR) T cell therapy for hematological malignancies has revolutionized anticancer therapy. However, CAR-T cells can cause immunity-related adverse events, such as cytokine release syndrome and neurotoxicity; the efficacy of CAR-T cell therapy also varies substantially with different patients. Poor understanding of plasma proteomic and metabolomic features during chimeric antigen receptor (CAR) T cell therapy may affect the exploitation of therapeutic potential in the management of hematological malignancies.
Methods: We longitudinally performed proteomic and metabolomic profiling of plasma from 20 patients with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL) on CAR-T cell therapy. Blood samples were collected from B-ALL patients at different time points, including before fludarabine/cyclophosphamide lymphodepletion (Pre-FC), Day 0 (D0), Day 7 (D7), Day 14 (D14), Day 28 (D28), and Peak-IL-6 during CAR-T cell therapy. The plasma samples from 22 age- and gender-matched healthy controls (HC) were used as controls.
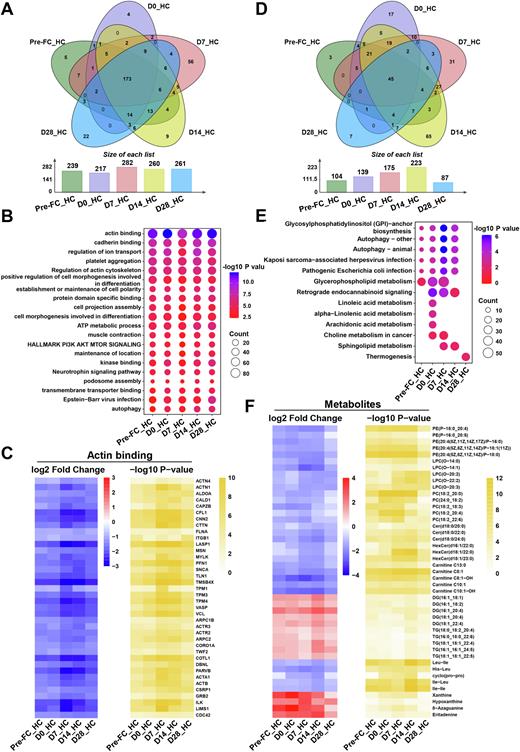
Results: Compared with HC, 239, 217, 282, 260 and 261 proteins were markedly different in terms of expression level in these five groups (Pre-FC, D0, D7, D14, D28 versus HC), respectively (Figure 1). The pathways of differentially expressed proteins (DEPs) were highly enriched in processes implicated in actin binding, cadherin binding, regulation of ion transport, platelet aggregation and regulation of actin cytoskeleton, with actin binding obtaining the most significant change (Figure 1). 104, 139, 175, 223 and 87 metabolites were significantly different in these five groups, respectively (Figure 1). Interestingly, the alterations of plasma metabolites became more extensive on the 14th day after CAR-T cell infusion, indicating that the change of metabolites may lag behind that of proteins. Furthermore, CAR-T cell therapy reshaped plasma proteome and metabolome with discrete kinetics, as represented by upregulated acute-phase response and triglycerides metabolism and downregulated actin cytoskeleton organization, retrograde endocannabinoid signaling and glycosylphosphatidylinositol (GPI)-anchor biosynthesis during the therapy. We also identified sets of covarying molecules that were involved in acute inflammatory response, epithelial mesenchymal transition, glutathione metabolism and tryptophan metabolism in concert with the dynamics of circulating IL-6 in patients with cytokines storm. Subgroup analysis further revealed numerous prognostic biomarkers, such as proteins related to allograft rejection, which were down-regulated in patients who achieved stable complete remission.
Conclusions: This study provided a dynamic landscape of plasma proteome and metabolome in B-ALL patients during CAR-T cell therapy and offered novel insights into CAR-T cell therapy-related toxicities and prognostic heterogeneity.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal